Overview
Control System
Automated Control
Sterilization
Vhp Generator
Manufacturing Materials
316L or PTFE
Production Capacity
5000pieces/Year
Package Size
340.00cm * 150.00cm * 200.00cm
Product Description
Marya Isolation Systems with Containment Technology for High-Potency Drug Production
The Marya Sterile Barrier Design is an advanced isolation system engineered to ensure maximum sterility and contamination control in aseptic drug manufacturing. Utilizing cutting-edge barrier technology, it provides a fully enclosed environment to protect sensitive pharmaceutical processes from microbial and particulate contamination. Designed for compliance with stringent GMP and regulatory standards, the system enhances product safety while optimizing operational efficiency.
Product Parameters
| Cleanliness | ISO4.8 |
| Temperature and humidity | Conventional 18-26ºC / 45%-65% (Customizable) |
| Differential pressure | Regular 20-50pa |
| Air flow rate | 0.36m/s-0.54m/s |
| Air flow type | Vertical one-way flow |
| Sterilization effect | log6 and above |
| Leakage rate | 0.5% |
Key Features

Customization and Integration:
- Modular Design: Integration of various modules for custom isolation needs.
- Accessories: Configurable with shelves, cleaning guns, and dehumidification.
- Control System: Automatic control of gas pressure, water, and oxygen content.
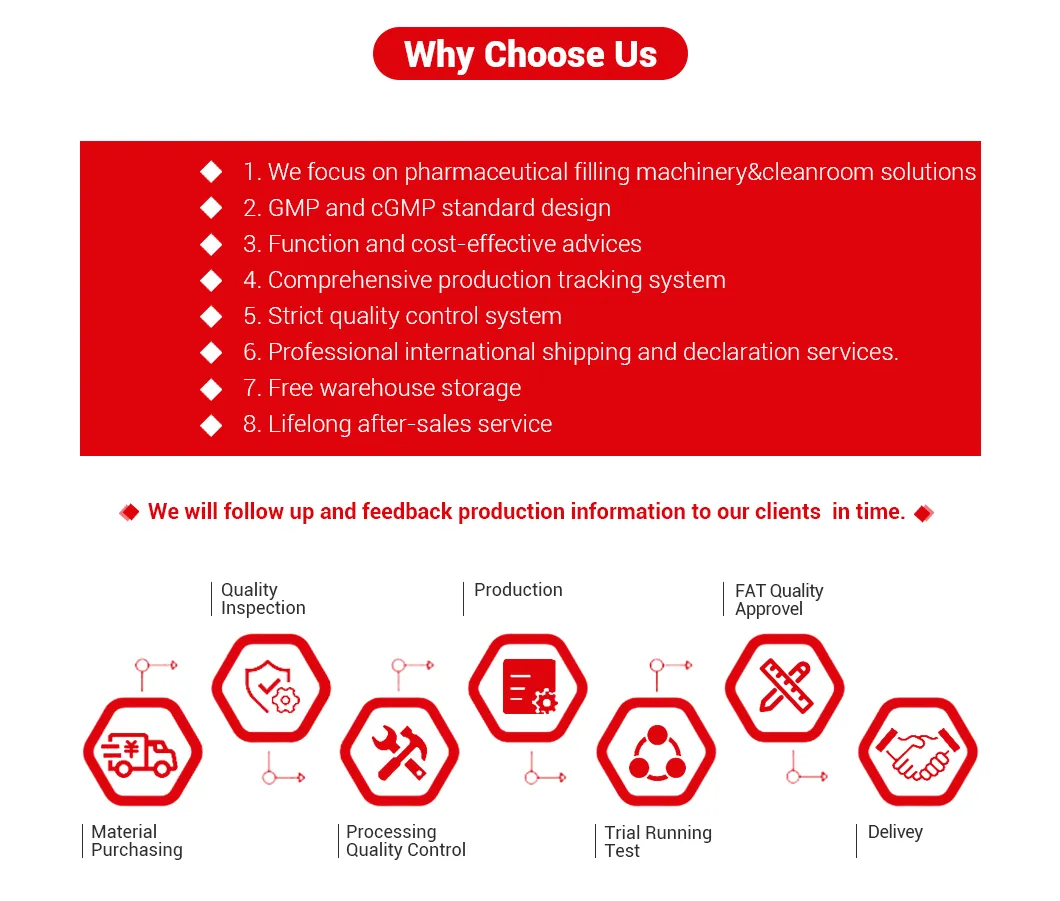
About Us & Why Choose Us

Workshop & Certificates
Product Category

Project Cases & Exhibition


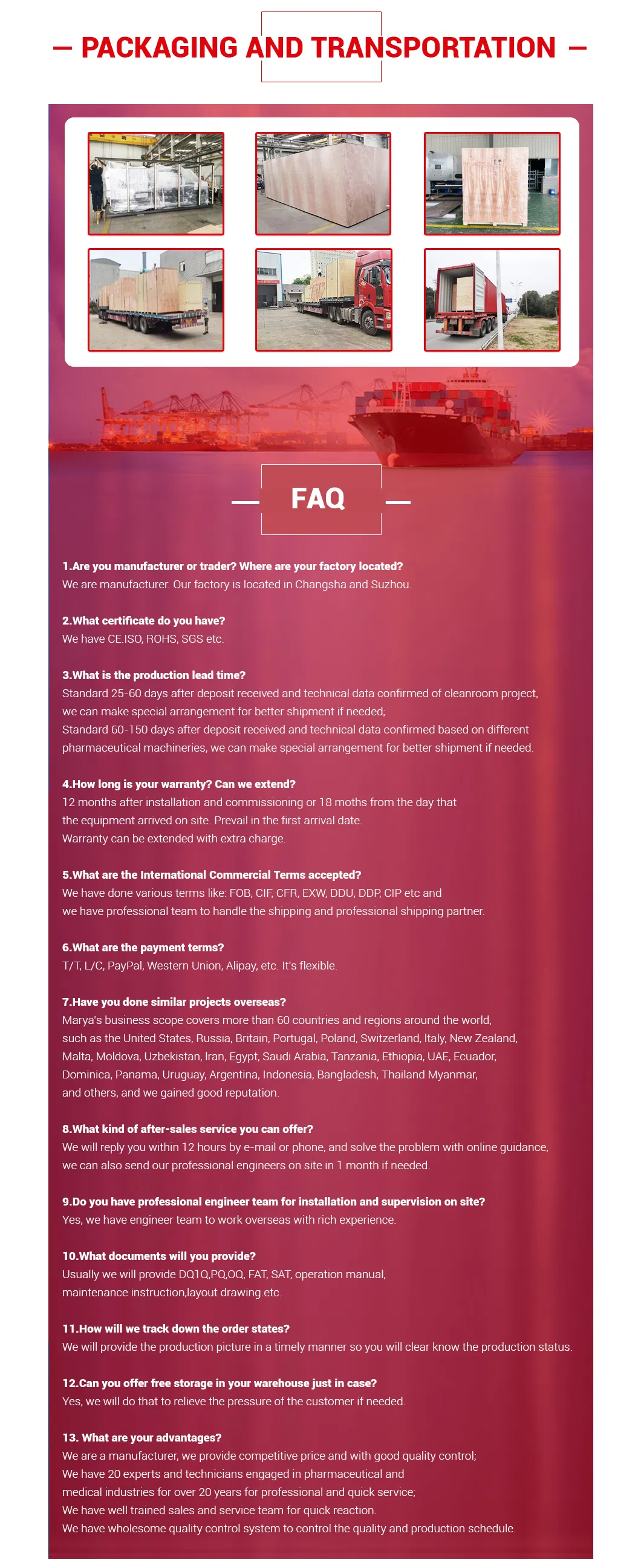
Packaging & Shipping
Frequently Asked Questions
Q1: What is the cleanliness level of the Marya Isolation System?
A1: The system achieves ISO 4.8 cleanliness, ensuring a superior aseptic environment for sensitive pharmaceutical production.
Q2: Can the sterilization effect reach log6?
A2: Yes, the system utilizing VHP (Vaporized Hydrogen Peroxide) sterilization can achieve a sterilization effect of log6 and above.
Q3: Is the isolation system customizable?
A3: Absolutely. We offer a modular design that allows for custom integration of shelves, dehumidification systems, and various cleaning tools based on your specific process needs.
Q4: What materials are used in manufacturing the system?
A4: The system is primarily constructed using 316L stainless steel or PTFE to ensure durability and GMP compliance.
Q5: How is the air flow managed inside the barrier?
A5: It utilizes a vertical one-way air flow type with a flow rate between 0.36m/s and 0.54m/s to maintain consistent environment stability.
Q6: What is the typical leakage rate for this equipment?
A6: The equipment is designed with high precision, maintaining a leakage rate as low as 0.5% for maximum containment safety.




























 Purr Pet
Purr Pet